- Research article
- Open Access
- Published:
Tomato SR/CAMTA transcription factors SlSR1 and SlSR3L negatively regulate disease resistance response and SlSR1L positively modulates drought stress tolerance
BMC Plant Biologyvolume14, Article number:286(2014)
Abstract
Background
The SR/CAMTA proteins represent a small family of transcription activators that play important roles in plant responses to biotic and abiotic stresses. SevenSlSR/CAMTAgenes were identified in tomato as tomato counterparts of SR/CAMTA; however, the involvement ofSlSRs/CAMTAsin biotic and abiotic stress responses is not clear. In this study, we performed functional analysis of theSlSR/CAMTAfamily for their possible functions in defense response against pathogens and tolerance to drought stress.
Results
Expression ofSlSRswas induced with distinct patterns byBotrytis cinereaandPseudomonas syringaepv.tomato(Pst) DC3000. Virus-induced gene silencing (VIGS)-based knockdown of eitherSlSR1orSlSR3Lin tomato resulted in enhanced resistance toB. cinereaandPstDC3000 and led to constitutive accumulation of H2O2, elevated expression of defense genes, marker genes for pathogen-associated molecular pattern-triggered immunity, and regulatory genes involved in the salicylic acid- and ethylene-mediated signaling pathways. Furthermore, the expression ofSlSR1LandSlSR2Lin detached leaves and whole plants was significantly induced by drought stress. Silencing ofSlSR1Lled to decreased drought stress tolerance, accelerated water loss in leaves, reduced root biomass and attenuated expression of drought stress responsive genes in tomato. The SlSR1 and SlSR3L proteins were localized in the nucleus of plant cells when transiently expressed inNicotiana benthamianaand had transcriptional activation activity in yeast.
Conclusions
VIGS-based functional analyses demonstrate that both SlSR1 and SlSR3L in the tomatoSlSR/CAMTAfamily are negative regulators of defense response againstB. cinereaandPstDC3000 while SlSR1L is a positive regulator of drought stress tolerance in tomato.
Background
Plants are vulnerable to various biotic and abiotic stresses but have evolved to equip with sophisticated signaling networks to precisely regulate defense response to unfavorable stresses. Upon perception of environmental stress, a set of early signaling events including changes in the cytosolic free calcium (Ca2+) concentration signatures (i.e. oscillations varying in cellular location, amplitude, duration or frequency) is often activated and integrated into different signaling pathways, which ultimately initiate transcriptional reprogramming leading to expression of a large set of stress-responsive genes [1]-[3]. Extensive biochemical and genetic studies have demonstrated that cellular Ca2+as a universal second messenger plays critical roles in regulating defense responses to diverse biotic and abiotic stresses [4]-[7].
Cellular Ca2+changes can be sensed and interpreted by calcium-binding proteins (CaBPs) including calmodulin (CaM), calcineurin B-like proteins and calcium-dependent protein kinases [8]-[10]. These CaBPs regulate cellular responses through two distinct pathways upon sensing different biotic and abiotic signals [8]-[10]. Firstly, CaBPs trigger rapid responses by direct binding to cytosolic target proteins and modulating their activity. Alternatively, CaBPs modulate indirect and relatively slow cellular responses by interacting with transcription factors to regulate gene expression. Recently, a class of CaM-binding transcription factors (CAMTA for CaM-binding transcription activator) has been identified in plants [11]-[14]. The CAMTA proteins, also called signal-responsive (SR) proteins, are present in all plant and animal species examined to date and are highly conserved in their protein structures. Typically, the SR/CAMTA proteins contain a CG-1 DNA-binding domain (binding to specificcis-elements in promoter regions of the target genes) at the N-terminus, a TIG domain (an immunoglobulin-like fold involved in nonspecific DNA binding), three ankyrin repeats (implicated in protein-protein interaction) and five putative CaM-binding motifs called as IQ motif [11]-[13]. Biochemical studies with the Arabidopsis AtSR1 and rice OsCBT proteins have identified the primary target of DNAcis-element for SR/CAMTA proteins as CGCG and CGTG motifs in promoter regions of the target genes [12],[14].
The SR/CAMTA proteins represent a small family of transcription activators in plants. For instance, six genes encoding for SR/CAMTA proteins were identified in Arabidopsis [11]. TheSR/CAMTAgenes were shown to be responsive to multiple abiotic and biotic stresses including cold, wounding, drought and pathogen attack, as well as to stress-related hormones like ethylene, auxin, methyl jasmonate (MeJA) and salicylic acid (SA) [11],[12],[15]-[18]. Recent genetic studies with loss-of-function and gain-of-function mutants have shown that members of the SR/CAMTA family play important roles in plant response to abiotic and biotic stresses. The ArabidopsisAtSR1knockout mutant showed enhanced disease resistance against multiple pathogens with different infection styles includingPseudomonas syringaepv.tomato(Pst),Botrytis cinereaandGolovinomyces cichoracearum[19]-[21] but decreased resistance against insect herbivores [22],[23]. By contrast, overexpression ofAtSR1conferred an increased susceptibility toPstDC3000,B. cinereaandG. cichoracearum[21],[24]. Similarly, the riceoscbtmutant exhibited significant resistance to blast fungal pathogenMagnaporthe griseaand leaf blight bacterial pathogenXanthomonas oryzaepv.oryzae[17]. On the other hand, it was recently shown that the Arabidopsis AtSR1 and AtSR2 also play important roles in regulating tolerance to low temperature [25],[26] and drought stress [27]. The function of SR/CAMTA proteins in plant biotic and abiotic stress response is achieved mainly through regulating expression of genes whose promoter regions contain the CGCG boxes [20],[21],[25],[27].
SevenSlSRs/CAMTAs(hereafter referred to asSlSRsfor convenience) genes were identified in tomato and were shown to be developmentally regulated during fruit development and ripening and induced by ethylene [28],[29]. Further detailed analysis revealed that theSlSRgenes showed differential expression patterns in tomato fruit in response to low temperature, mechanical injury, infection of the necrotrophic fungal pathogenB. cinerea, and treatments with the signaling molecules SA and MeJA [30.]. However, direct genetic evidence supporting the involvement of the tomatoSlSRsin biotic and abiotic stress responses is still lacking. In the present study, we investigated the possible functions ofSlSRsin disease resistance and drought stress tolerance using virus-induced gene silencing (VIGS) approach. Our VIGS-based functional analyses demonstrate that both SlSR1 and SlSR3L are negative regulators of defense response againstB. cinereaandPstDC3000 while SlSR1L is a positive regulator of drought stress response.
Results
Expression patterns ofSlSRsin response to pathogen infection
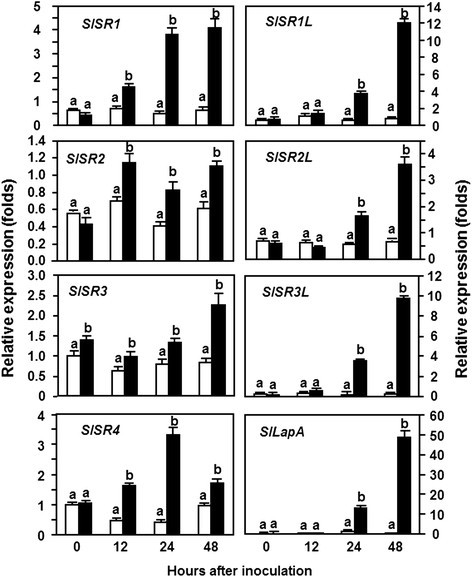
To explore the possible functions ofSlSRsin defense response against pathogen infection, we analyzed the expression patterns ofSlSRgenes in tomato plants after infection with different pathogens,B. cinerea, a necrotrophic fungal pathogen causing grey mold disease, andPstDC3000, a (hemi) biotrophic bacterial pathogen causing bacterial leaf spot disease. In analysis of expression patterns ofSlSRsin response to infection ofB. cinerea, leaf samples collected from the whole plant inoculation assays were used and the expression pattern ofSlLapA, a defense gene regulated by the JA/ET-mediated signaling pathway [31], was monitored to confirm the efficiency of the inoculation procedure. As shown in Figure1, the expression level ofSlLapAinB. cinerea-inoculated plants increased significantly, leading to 50 folds of increase at 24 hr post inoculation (hpi) and >300 folds of increase at 48 hpi relative to those in the mock-inoculated plants, suggesting that our inoculation assays were appropriate for further analysis of the expression patterns ofSlSRsin response toB. cinerea.Data from repeated qRT-PCR analyses revealed that the expression of almost allSlSRswas induced by infection ofB. cinereaand the induced expression ofSlSRswas evident after 24 hpi and showed distinct patterns (Figure1). The expression levels ofSlSR1,SlSR1L,SlSR2LandSlSR3Lwere induced significantly byB. cinerea, leading to increases of 8, 8, 3 and 15 folds at 24 hpi and of 8, 20, 7 and 90 folds at 48 hpi, respectively, over those in the mock-inoculated plants (Figure1). The expression level ofSlSR4in theB. cinerea-inoculated plants showed 3-fold increase at 12 hpi, peaked with 7-fold of increase at 24 hpi and then declined to the level in the mock-inoculated plants (Figure1). By contrast, the expression levels ofSlSR2andSlSR3in theB. cinerea-inoculated plants exhibited slight increases with less than 2 folds as compared with those in the mock-inoculated plants (Figure1). These results indicate that the expression ofSlSRscould be induced with distinct patterns byB. cinereaand that the expression of mostSlSRgenes except forSlSR2andSlSR3was highly responsive to infection ofB. cinerea.
Expression ofSlSRsin response to infection byB. cinerea.Four-week-old plants were inoculated by foliar spraying with spore suspension (2 × 105spores/ml) ofB. cinerea(filled bars) or with same volume of buffer as a mock inoculation control (open bars). Leaf samples were collected at indicated time points after inoculation for analysis ofSlSRexpression by qRT-PCR using gene-specific primers. Relative expression levels were calculated after normalization with actin transcript values. Data presented are the means ± SD from three independent experiments and different letters above the columns indicate significant differences atp?
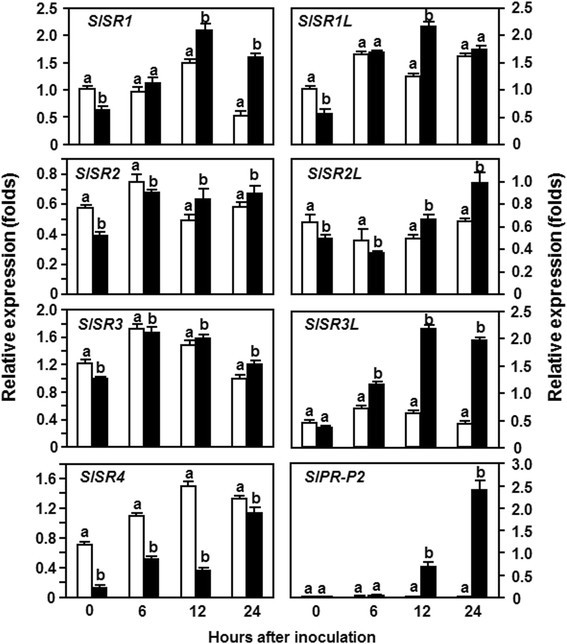
We next analyzed the expression patterns ofSlSRsin response to a virulent strain ofPstDC3000. In these experiments, the expression pattern ofSlPR-P2, a defense gene regulated by the SA-mediated signaling pathway [31], was examined to confirm the efficiency of the inoculation procedure. As shown in Figure2, the expression level ofSlPR-P2in thePstDC3000-inoculated plants increased at 6 hpi and showed 50 and >300 folds of increase at 12 and 24 hpi, over those in the mock-inoculated plants, confirming that the effectiveness of the inoculation procedure was satisfied for further analysis of the expression patterns ofSlSRsin response toPstDC3000. The expression levels ofSlSR1L,SlSR2,SlSR2LandSlSR3in thePstDC3000-inoculated plants were similar to those in the mock-inoculated plants, indicating that their expression was not responsive to infection ofPstDC3000 (Figure2). However, the expression levels ofSlSR1andSlSR3Lwere significantly induced byPstDC3000 (Figure2). A 3-fold increase of the expression ofSlSR1in thePstDC3000-inoculated plants was observed at 24 hpi but no significant increase in the expression level ofSlSR1in thePstDC3000-inoculated plants was observed within the first 12 hpi, as compared with those in the mock-inoculated plants (Figure2). The expression level ofSlSR3Lin thePstDC3000-inoculated plants exhibited 2-fold and 4-fold increases at 12 hpi and 24 hpi, respectively, relative to those in the mock-inoculated plants (Figure2). Interestingly, the expression ofSlSR4in thePstDC3000-inoculated plants was suppressed byPstDC3000 during the first 12 hpi and this suppression ofSlSR4expression was very quick as the expression level ofSlSR4in thePstDC3000-inoculated plants decreased by approximately 4 folds relative to that in the mock-inoculated plants (Figure2). The expression level ofSlSR4in thePstDC3000-inoculated plants restored to the level in the mock-inoculated plants (Figure2). These results indicate that the expression ofSlSRscould be induced with distinct patterns byPstDC3000 and that the expression ofSlSR1andSlSR3Lwas induced but the expression ofSlSR4was suppressed byPstDC3000.
Expression ofSlSRsin response to infection byP. syringaepv.tomatoDC3000.Four-week-old plants were inoculated by vacuum infiltration withP. syringaepv.tomatoDC3000 (OD600?=?0.0002) (filled black bars) or with 10 mM MgCl2solution as a mock inoculation control (open bars). Leaf samples were collected at indicated time points after inoculation for analysis ofSlSRexpression by qRT-PCR using gene-specific primers. Relative expression folds were calculated after normalization with actin transcript values. Data presented are the means ± SD from three independent experiments and different letters above the columns indicate significant differences atp?
Silencing of SlSR1 and SlSR3L conferred an increased resistance to B. cinerea and Pst DC3000
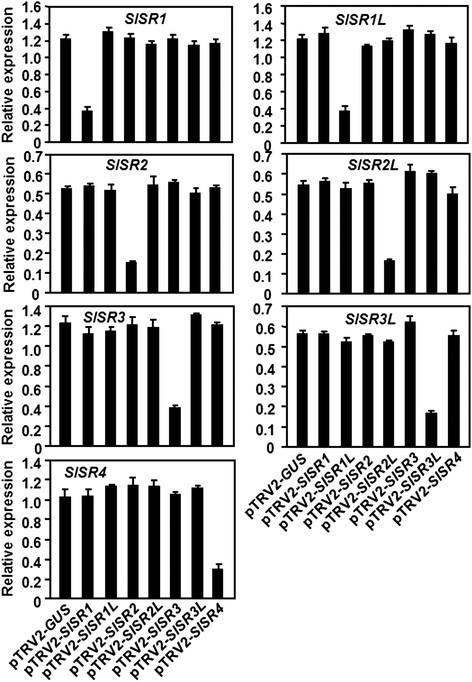
To explore the possible functions ofSlSRsin plant defense response, we used the TRV-based VIGS system [32] to knockdown the expression levels ofSlSRgenes in tomato plants and compared the phenotypes and severity of diseases caused byB. cinereaandPstDC3000, respectively. In our VIGS study, we chose sequences that encode for the highly diverged regions in the SlSR proteins to avoid interference with expression of other non-targetSlSRgenes when one targetSlSRgene was attempted to be silenced (Additional file1). Silencing efficiency and specificity were assessed by qRT-PCR analyzing the transcript abundance of the targetSlSRgene in the silenced and non-silenced pTRV2-GUS-infiltrated control plants. As shown in Figure3的转录水平SlSRgene in the corresponding silenced plants were significantly reduced, leading to the silencing efficiency of 70–75% in standard VIGS experiments, as compared with those in the pTRV2-GUS-infiltrated plants. By contrast, the transcript levels of eachSlSRgene in those plants that were silenced for one of the other sixSlSRgenes were comparable to the level in the pTRV2-GUS-infiltrated control plants (Figure3), indicating that silencing only occurred for the targeted gene but not for the other non-targetedSISRgenes. Thus, the silencing efficiency and specificity under our experiment conditions were satisfied for further study and all the subsequent experiments were performed only on those pTRV2-SlSR-infiltrated plants with high levels of silencing efficiency (>70%).
Silencing efficiency and specificity for target genes in silenced plants.Two-week-old tomato seedlings were infiltrated with agrobacteria carrying pTRV2-SlSRs or pTRV2-GUS constructs and leaf samples were collected from pTRV2-SlSRs- and pTRV2-GUS-infitlrated plants at 4 weeks after agroinfiltration. Expression levels of eachSlSRgenes in targeted and nontargetedSlSRgenes-silenced and non-silenced plants were analyzed by qRT-PCR and data obtained were normalized with actin transcript values. Data presented are the means ± SD from three independent experiments.
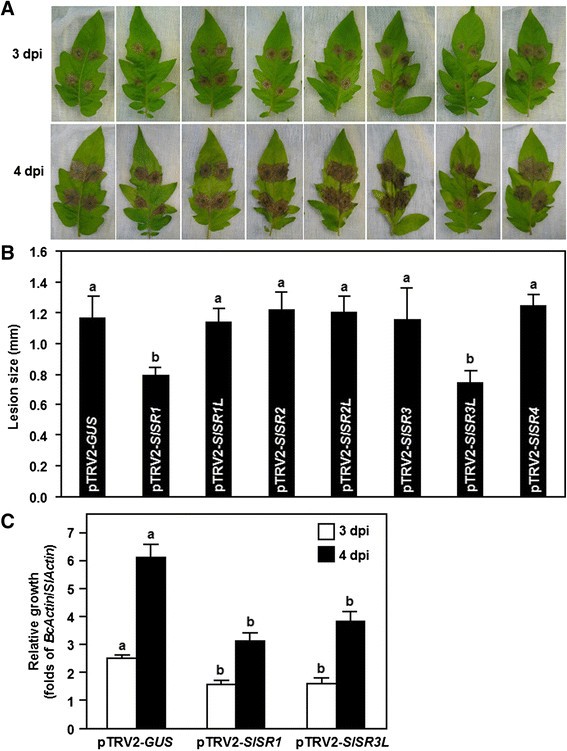
We first examined the possible roles ofSlSRsin resistance againstB. cinereaby challenging the pTRV2-SlSRs-infiltrated plants with spore suspension ofB. cinereaand comparing the disease severity andin plantafungal growth with those in pTRV-GUS-infiltrated non-silenced plants. In our detached leaf assays,B. cinerea-caused lesions on detached leaves from the pTRV2-SlSR1L-, pTRV2-SlSR2-, pTRV2-SlSR2L-, pTRV2-SlSR3- and pTRV2-SlSR4-infiltrated plants were similar to the lesions on the detached leaves from pTRV2-GUS-infilrtratd plants (Figure4A and B), suggesting thatSlSR1L,SlSR2,SlSR2L,SlSR3andSlSR4may not be involved in disease resistance againstB. cinerea.However,B. cinerea-caused lesions on detached leaves from the pTRV2-SlSR1- and pTRV2-SlSR3L-infiltrated plans developed slowly and were still separated, as compared with the large merged lesions on leaves from the pTRV2-GUS-infiltrated plants, at 4 days after inoculation (dpi) (Figure4A). At 4 dpi, the lesion sizes on detached leaves from the pTRV2-SlSR1- and pTRV2-SlSR3L-infiltrated plants were significantly reduced, leading to a reduction of approximately 35%, as compared with that of the pTRV2-GUS-infiltrated plants (Figure4B). We further analyzed and compared thein plantafungal growth in the pTRV2-SlSR1-, pTRV2-SlSR3L- and pTRV2-GUS-infiltrated plants after inoculation by foliar spraying with spore suspension ofB. cinereain whole plant inoculation experiments. qRT-PCR analysis of the transcript levels of theB. cinereaactin geneBcActin, which was used as an indicative of the rate of fungal growthin planta, showed that the fungal growth in the pTRV2-SlSR1- and pTRV2-SlSR3L-infilrated plants were significantly suppressed, resulting in reductions of 35–51% at 3 and 4 dpi, as compared with those in the pTRV2-GUS-infiltrated plants (Figure4C). These results indicate that theSlSR1- andSlSR3L-silenced plants were more resistant toB. cinereainfection than the pTRV2-GUS-infiltrated plants. Taken together, these data demonstrate that silencing ofSlSR1orSlSR3Lresulted in increased resistance againstB. cinereaand thus both SlSR1 and SlSR3L may act as negative regulators of disease resistance againstB. cinerea.
Silencing of eitherSlSR1orSlSR3Lresulted in enhanced disease resistance toB. cinerea.Two-week-old tomato seedlings were infiltrated with agrobacteria carrying pTRV2-SlSRs or pTRV2-GUS constructs and leaves were detached from pTRV2-GUS- or pTRV2-SlSR-infiltrated plants at 3 weeks after VIGS infiltration. Inoculation withB. cinereawas done by dropping 5 ?l of spore suspension (1 × 105spores/ml).(A)Disease symptom on detached leaves at 3 days after inoculation.(B)Lesion size in leaves of the pTRV2-GUS- or pTRV2-SlSR-infiltrated plants at 4 days after inoculation. At least 10 leaves from ten individual plants were used for each experiment.(C)Growth ofB. cinereain inoculated plants from whole plant inoculation experiments. Fungal growthin plantawas assumed at 3 and 4 days after inoculation by qRT-PCR analyzing the transcript level ofB. cinerea BcActinAgene usingSlActingene as an internal control. Relative fungal growth was shown as folds of transcript levels ofBcActincompared toSlActin.Data presented are the means ± SD from three independent experiments and different letters above the columns indicate significant differences atp?
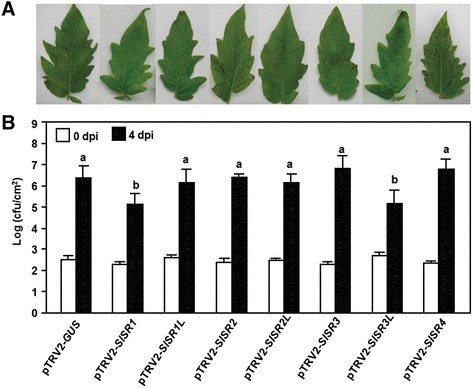
We next explored whetherSlSRshave functions in resistance againstPstDC3000. Disease phenotypes and bacterial growthin plantawere compared between the pTRV2-SlSRs- and pTRV2-GUS-infiltrated plants after inoculation with a virulent strain ofPstDC3000. At 4 dpi, the pTRV2-GUS-infiltrated plants showed typical bacterial speck disease symptoms, including necrotic lesions surrounded by chlorosis (Figure5A). The disease severity and bacterial growthin plantain inoculated pTRV2-SlSR1L-, pTRV2-SlSR2-, pTRV2-SlSR2L-, pTRV2-SlSR3- and pTRV2-SlSR4-infiltrated plants were comparable to those in the pTRV2-GUS-inflitrated plants (Figure5A and B), suggesting thatSlSR1L,SlSR2,SlSR2L,SlSR3andSlSR4are not involved in disease resistance toPstDC3000. By contrast, the pTRV2-SlSR1- and pTRV2-SlSR3L-infiltrated plants showed very weak visible symptoms of disease caused byPstDC3000, as compared with that in the pTRV2-GUS-infiltrated plants (Figure5A). This reduced disease symptoms on leaves of the pTRV2-SlSR1- and pTRV2-SlSR3L-infiltrated plants were coincided with relatively low levels of bacterial growthin plantaafter inoculation withPstDC3000. At 4 dpi, the bacterial populations in leaves of the pTRV2-SlSR1- and pTRV2-SlSR3L-infiltrated plants were 2.8 × 105and 2.2 × 105CFU/cm2, leading to a reduction of 10-fold, as compared to that in leaves of the pTRV-GUS-infiltrated plants (2.5 × 106CFU/cm2) (Figure5B). These results demonstrate that silencing of eitherSlSR1orSlSR3Lled to increased resistance againstPstDC3000 and thus both of SlSR1 and SlSR3L are also negative regulators of disease resistance toPstDC3000.
Silencing of eitherSlSR1orSlSR3Lled to enhanced resistance toP. syringaepv.tomatoDC3000.Two-week-old tomato seedlings were infiltrated with agrobacteria carrying pTRV2-SlSRs or pTRV2-GUS constructs and the pTRV2-GUS- and pTRV2-SlSRs-infiltrated plants were inoculated by vacuum infiltration with infiltration withP. syringaepv.tomatoDC3000 (OD600?=?0.0002) at 3 weeks after VIGS infiltration.(A)Representative symptom of disease caused byP. syringaepv.tomatoDC3000 at 4 days after inoculation.(B)Bacterial growth in inoculated leaves of pTRV2-GUS- and pTRV2-SlSRs-infiltrated plants. Leaf samples were collected at 0 and 4 days after inoculation and bacterial growth was measured. Data presented are the means ± SD from three independent experiments and different letters above the columns indicate significant differences atp?
Silencing of SlSR1 and SlSR3L resulted in constitutive defense response
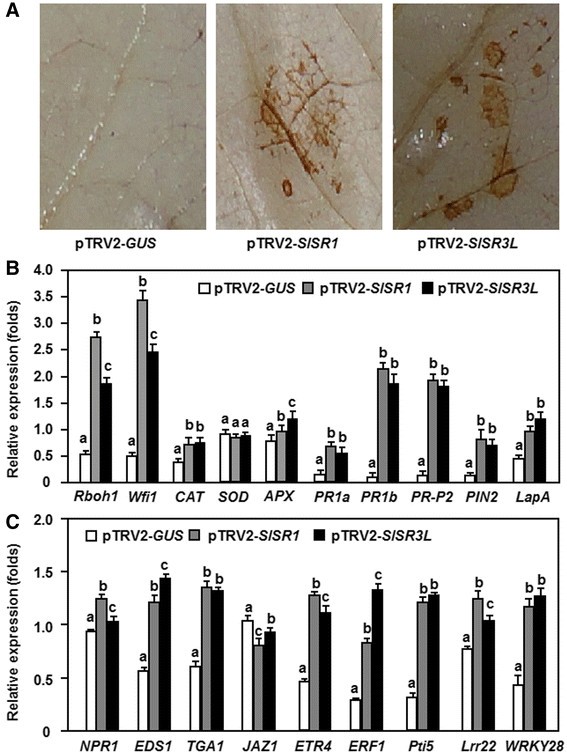
The Arabidopsissr1mutant plants were previously found to display chlorosis and constitutive expression of defense genes under lower temperature, indicating that loss of SR1 function in Arabidopsis led to constitutive defense response [20]. To examine whether silencing ofSlSR1orSlSR3Lcould also confer constitutive defense responses, we analyzed and compared the accumulation of H2O2and expression of genes involved in different signaling pathways and defense response between the pTRV2-SlSR1- and pTRV2-SlSR3L-infiltrated plants and the pTRV2-GUS-infiltrated plants. Results from 3,3-diaminobenzidine (DAB) staining ofin situH2O2accumulation showed that significant brown precipitates, representing the accumulation of H2O2,很容易和清晰地观察到叶子的e pTRV2-SlSR1- and pTRV2-SlSR3L-infiltrated plants without infection of pathogen, while no significant brown precipitate was seen in leaves of the pTRV2-GUS-infiltrated plants (Figure6A). These results indicate that silencing of eitherSlSR1orSlSR3Lresulted in constitutive accumulation of H2O2in tomato plants. To explore the possible mechanism for the constitutive accumulation of H2O2in theSlSR1- andSlSR3L-silencedd plants, we analyzed and compared the expression of genes encoding for NADPH oxidases, which are plasma membrane-localized ROS generating enzymes [33], and for catalases (CAT), superoxide dismutases (SOD) and ascorbate peroxidases (APX), which are involved in scavenging of ROS, in the pTRV2-SlSR1- and pTRV2-SlSR3L-infiltrated plants. As shown in Figure6B, the expression levels ofRboh1andWfi1, two genes for NADPH oxidases, in the pTRV2-SlSR1- and pTRV2-SlSR3L-infiltrated plants were significantly elevated, giving increases of 5?~?7-fold forRboh1and 4?~?5-fold forWfi1, as compared with those in the pTRV2-GUS-infiltrated plants. Similarly, the expression levels ofCATandAPXin the pTRV2-SlSR1- and pTRV2-SlSR3L-infiltrated plants were also increased as compared with those in the pTRV2-GUS-infiltrated plants (Figure6B). By contrast, no significant difference was observed in the expression level ofSODbetween the pTRV2-SlSR1- and pTRV2-SlSR3L-infiltrated plants and the pTRV2-GUS-infiltrated plants (Figure6B). These results indicate that the constitutive accumulation of H2O2in theSlSR1- andSlSR3L-silenced plants might be attributed to the increased ROS generating ability resulted from the high level of expression of the NADPH oxidases.
Silencing of eitherSlSR1orSlSR3Lactivated constitutive defense response.Two-week-old seedlings were infiltrated with agrobacteria carrying pTRV2-SlSR1, pTRV2-SlSR3L or pTRV2-GUS constructs and leaf samples were collected at 2 weeks after VIGS infiltration for detection of H2O2accumulation and analysis of expression of signaling- and defense-related genes.(A)Accumulation of H2O2in leaves of the pTRV2-SlSR1- and pTRV2-SlSR3L-infiltrated plants.(B)Expression of defense-related genes and ROS production-related genes in the pTRV2-SlSR1- and pTRV2-SlSR3L-infiltrated plants.(C)Expression of genes involved in different defense signaling pathways in the pTRV2-SlSR1- and pTRV2-SlSR3L-infiltrated plants. Data presented are the means ± SD from three independent experiments and different letters above the columns indicate significant differences atp?
We next examined whether silencing ofSlSR1orSlSR3Lled to constitutive expression of defense genes. The expression levels of five representative defense genes that are regulated through different defense signaling pathways and three marker genes for pathogen-associated molecular patterns-triggered immunity (PTI) were analyzed and compared between the pTRV2-SlSR1- and pTRV2-SlSR3L-infiltrated plants and the pTRV2-GUS-infiltrated plants. As shown in Figure6B, the expression levels ofPR1a,PR1bandPR-P2, which are thought to be regulated by the SA-mediated signaling pathway [31], and ofPIN2andLapA,这被认为是由农协/ ET signaling pathway [31], were increased significantly in the pTRV2-SlSR1- and pTRV2-SlSR3L-infiltrated plants, leading to 8?~?25 folds of increases for thePR1a,PR1bandPR-P2genes and 1?~?11 folds of increases for thePIN2andLapAgenes, as compared with those in the pTRV2-GUS-infiltrated plants. Similarly, the expression levels ofPti5,Lrr22andWRKY28, three PTI marker genes in tomato [34],[35], in the pTRV2-SlSR1- and pTRV2-SlSR3L-infiltrated plants were significantly higher than those in the pTRV2-GUS-infiltrated plants, resulting in increases of 0.5?~?3.0 folds in the expression levels (Figure6C). These data demonstrate that silencing of eitherSlSR1orSlSR3Lactivates constitutively the expression of the defense and PTI genes, leading to constitutive defense response in theSlSR1- andSlSR3L-silenced plants.
To determine whether silencing ofSlSR1andSlSR3Lactivates defense signaling pathways, we further analyzed and compared the expression levels of the key genes encoding important components involved in the SA- and JA/ET-mediated signaling pathways between the pTRV2-SlSR1- and pTRV2-SlSR3L-infiltrated plants and the pTRV2-GUS-infiltrated plants. The expression levels ofNPR1,EDS1andTGA1, known to be critical components in the SA-mediated signaling pathway [36], in the pTRV2-SlSR1- and pTRV2-SlSR3L-infiltrated plants were significantly increased, especially for the expression levels of theEDS1andTGA1genes, showing 1-fold increase relative to those in the pTRV2-GUS-infiltrated plants (Figure6C). Similarly, the expression levels ofETR4andERF1, known to be associated with ET signaling pathway [37],[38], in the pTRV2-SlSR1- and pTRV2-SlSR3L-infiltrated plants showed 1?~?2-fold increase over those in the pTRV2-GUS-infiltrated plants (Figure6C). By contrast, the expression level ofJAZ1, known to be associated with JA signaling pathway [39], in the pTRV2-SlSR1- and pTRV-SlSR3L-infiltrated plants was reduced as compared with those in the pTRV2-GUS-infiltrated plants (Figure6C). These results suggest that silencing of eitherSlSR1orSlSR3Lcan activate both the SA- and ET-mediated signaling pathways but suppress the JA-mediated signaling pathway in theSlSR1- andSlSR3L-silenced plants.
Expression patterns ofSlSRsin response to drought stress and ABA
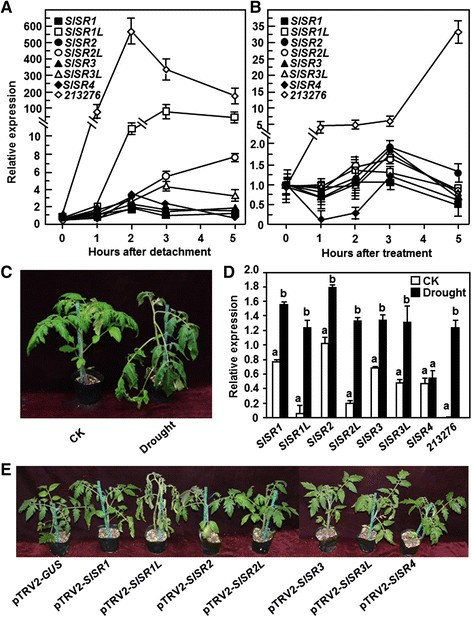
To explore the possible involvement ofSlSRsin drought stress response, we first examined whether the expression ofSlSRs可能会引起干旱胁迫和abscisci交流吗id (ABA) treatment. Two different methods, the detached leaf and the whole plant assays, were adapted to analyze the expression ofSlSRsin response to drought stress. In these experiments, a previously reported drought stress-responsive gene,SGN-213276[40], was included to confirm the efficiency of the drought stress treatment. The expression level ofSGN-213276was markedly increased after drought stress treatment, leading to increases of >100-fold at 5 hr after treatment in the detached leaf assays (Figure7A) and at 7 days after treatment in the whole plant assays (Figure7D), indicating that the experiments for drought stress assays were satisfied for further analyzing the expression of theSlSRgenes. In the detached leaf assays, as compared with the expression levels of the corresponding genes in the water-saturated detached leaves, no significant change in the expression levels ofSlSR1,SlSR2,SlSR3andSlSR4was observed over a period of 5 hr after detachment (Figure7A), whereas a maximal increase of 4?~?8-fold in the expression levels ofSlSR2LandSlSR3Lwas observed during 3–5 hr after treatment (Figure7A). By contrast, the expression kinetic ofSlSR1Lunder drought stress condition was similar to that of theSGN-213276gene (Figure7A). The expression level ofSlSR1Lin the drought stress treated leaves started to increase at 2 hr after detachment and exhibited approximately an increase of 50?~?90 folds as compared with that in the water-saturated leaves (Figure7A). In the whole plant assays, the drought stressed plants exhibited clear wilting symptom at 7 days after withholding water whereas the normally watered plants did not show any wilting symptom (Figure7C). qRT-PCR analyses revealed that the expression ofSlSRsexceptSlSR4was induced by drought stress. The expression levels ofSlSR1,SlSR2,SlSR3andSlSR3Lin the drought stressed plants showed a slight increase, leading to an increase of approximately 1 fold or less over those in the normally watered plants (Figure7D). However, the expression levels ofSlSR1LandSlSR2Lin the drought stressed plants increased significantly as compared with those in the normally watered plants, resulting in increases of 20 folds and 7 folds, respectively (Figure7D). Taken together, these results suggest that someSlSRsespeciallySlSR1Lare responsive to drought stress. We also analyzed whether the expression ofSlSRscould be induced by exogenous application of ABA, a well-known hormone involved in drought stress response [41]. In these experiments, the expression level of the drought stress-responsive geneSGN-213276in the ABA-treated plants was markedly increased, leading to increases of 5?~?34-fold over those in the control plants (Figure7B); however, the expression levels ofSlSRsin the ABA-treated plants showed changes with less 2 folds as compared with those in the control plants (Figure7B), indicating that exogenous ABA did not affect the expression ofSlSRs.
Expression patterns ofSlSRsin response to drought stress and ABA treatment and phenotypes of theSlSRs-silenced plants under drought condition. (A)Expression ofSlSRsin detached leaves under drought stress. Fully expanded leaves were detached from four-week-old plants and subjected to drought stress treatment by placing on lab blench or water-saturated filter papers in Petri dishes as a control and samples were collected at different time points as indicated.(B)Expression ofSlSRsin detached leaves after ABA treatment. Four-week-old plants were treated by foliar spraying with ABA solution (100 ?M) or water as control and leaf samples were collected at different time points as indicated. Relative expression levels of theSlSRgenes in the treated plants were shown as folds of the expression levels in the control plants after normalization with actin transcript values.(C)and(D)Expression ofSlSRsin leaves of plants under drought stress. Four-week-old plants were treated for drought stress by stopping watering for a period or watered normally as controls and leaf samples were collected at 7 days after treatment when wilting symptom appeared(C).Total RNA was extracted and used for qRT-PCR analysis of expression ofSlSRs(D).Relative expression levels of theSlSRgenes in the treated and control plants were shown as folds of the actin transcript values.(E)Phenotypes of theSlSRs-silenced plants under drought condition. Two-week-old seedlings were infiltrated with agrobacteria carrying pTRV2-SlSRs or pTRV2-GUS constructs and 3 weeks later the pTRV2-GUS- and pTRV2-SlSRs-infiltrated plants were subjected to drought stress by stopping watering for 10 days. Data presented in(A),(B)and(D)are the means ± SD from three independent experiments and different letters above the columns in(D)indicate significant difference atp< 0.05 level.
Silencing of SlSR1L reduced drought tolerance in tomato
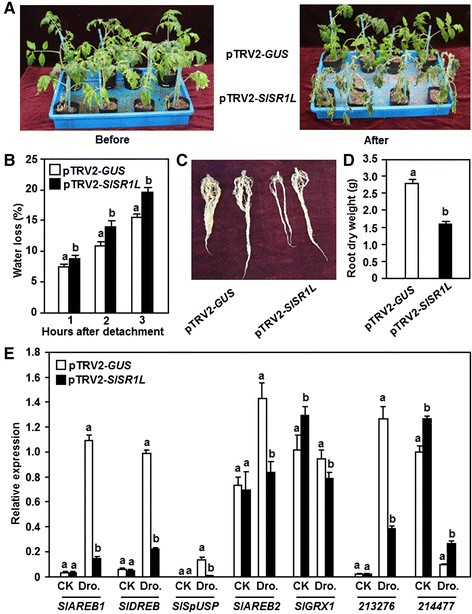
VIGS assays were performed to explore the involvement of individualSlSRsin drought stress response. For this purpose, we stopped watering for 7–10 days to compare the phenotype between the pTRV2-SlSRs- and pTRV2-GUS-inflitrated plants. In repeated experiments, silencing ofSlSR1,SlSR2,SlSR2L,SlSR3,SlSR3LorSlSR4did not affect drought stress response over an experimental period of 10 days (Figure7E), indicating that theseSlSRgenes may not be involved in drought stress response. However, the pTRV-SlSR1L-infiltrated plants showed significant wilting symptom at 7 days after drought stress treatment as compared with that of the pTRV2-GUS-infiltrated plants (Figure7E), implying a role forSlSR1Lin drought stress tolerance. The pTRV2-SlSR1L-infiltrated plants grew well as the pTRV2-GUS-infiltrated plants before withholding water, but they were easier to appear wilting symptom after stopping watering and their leaves became curly and the plants wilted at 7 days after withholding water (Figure8A). This result indicates that silencing ofSlSR1Lattenuated the drought stress tolerance in tomato. To explore the possible mechanism for the reduced drought stress tolerance in theSlSR1L-silenced plants, we first analyzed and compared the physiological and morphological changes between the pTRV2-SlSR1L- and pTRV2-GUS-infiltrated plants before and after drought stress treatment. The rate of water loss in leaves from the pTRV2-SlSR1L-infiltrated plants was higher than those in leaves from the pTRV2-GUS-infiltrated plants during the first 3 hr after detachment (Figure8B), indicating that silencing ofSlSR1Laccelerated water loss in leaves. The pTRV2-SlSR1L-infiltrated plants had smaller root system as compared to the pTRV2-GUS-infiltrated plants (Figure8C). Similarly, dry weights of the roots from the pTRV2-SlSR1L-infiltrated plants were significantly lower than that of the roots from the pTRV2-GUS-infiltrated plants, resulting in a reduction of approximately 40% (Figure8D). Furthermore, we also analyzed and compared the expression of some previously reported drought stress-responsive genesSlAREB1[42],SlAREB2[42],SlDREB[43],SlSpUSP[44],SlGRX1[45],SGN-213276[40] andSGN-214777[40] in the pTRV2-SlSR1L- and pTRV2-GUS-infiltrated plants before and after drought stress treatment. Before drought stress treatment, the expression levels ofSlAREB1,SlAREB2,SlDREB,SlSpUSPandSGN-213276in the pTRV2-SlSR1L-infiltrated plants were comparable to those in the pTRV2-GUS-infiltrated plants, whereas the expression levels ofSlGRX1andSGN-214777the pTRV2-SlSR1L-infiltrated plants showed a slight increase as compared with those in the pTRV2-GUS-infiltrated plants (Figure8E). At 10 days after drought stress treatment, the expression levels ofSlAREB1,SlDREB,SlSpUSP,SlAREB2andSGN-213276in the pTRV2-GUS-infiltrated plants increased significantly as compared with those in the normally watered control plants (Figure8E). After drought stress treatment, however, the expression levels ofSlAREB1,SlDREB,SlSpUSP,SlAREB2andSGN-213276in the pTRV2-SlSR1L-infiltrated plants decreased markedly as compared with those in the pTRV2-GUS-infiltrated plants (Figure8E). The expression level ofSGN-214777decreased in the pTRV2-GUS-infiltrated plants as compared with that in the normally watered plants but increased significantly in the pTRV2-SlSR1L-infiltrated plants as compared with that in the pTRV2-GUS-infiltrated plants after drought stress treatment (Figure8E).
Silencing ofSlSR1Lled to reduced drought stress tolerance.Two-week-old tomato seedlings were infiltrated with agrobacteria carrying pTRV2-SlSR1L or pTRV2-GUS constructs and 3 weeks later the pTRV2-GUS- and pTRV2-SlSR1L-infiltrated plants were subjected to drought stress by stopping watering.(A)Phenotype of the pTRV2-GUS- and pTRV2-SlSR1L-infiltrated plants before and after treatment of drought stress.(B)Rates of water loss in detached leaves of the pTRV2-GUS- and pTRV2-SlSR1L-infiltrated plants.(C)Root system of the pTRV2-GUS- and pTRV2-SlSR1L-infiltrated plants. The intact root systems are shown from one representative pTRV2-GUS- or pTRV2-SlSR1L-infiltrated plant.(D)Dry weights of roots from the pTRV2-GUS- and pTRV2-SlSR1L-infiltrated plants.(E)一些drought-responsive基因的表达pTRV2-GUS- and pTRV2-SlSR1L-infiltrated plants before and after treatment of drought stress. Data presented in(B),(C)and(E)are the means ± SD from three independent experiments and different letters above the columns indicate significant differences atp?
SlSR1 and SlSR3L are localized in nucleus and have transactivation activity
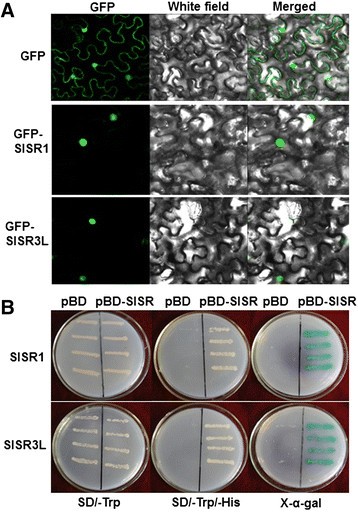
因为SlSR1的重要性和SlSR3L defense response againstB. cinereaandPstDC3000, we investigated the biochemical characteristics of these two SlSR proteins. Firstly, we examined the subcellular localization of SlSR1 and SlSR3L using a transient expression approach. We transiently expressed SlSR1 and SlSR3L in leaves of 4-week-oldN. benthamianaplants by infiltration with agrobacteria carrying pFGC-Egfp-SlSR1, pFGC-Egfp-SlSR3L or pFGC-Egfp constructs and GFP was observed at 2 days after agroinfiltration. As shown in Figure9A, the SlSR1-GFP and SlSR3L-GFP fusions accumulated exclusively in the nucleus ofN. benthamiana细胞,而我独自GFP蛋白积累n both the cytoplasm and the nucleus, demonstrating that both SlSR1 and SlSR3L proteins are localized in the nucleus of cells. Furthermore, we also examined whether the SlSR1 and SlSR3L proteins had transactivation activity using a yeast assay system. As shown in Figure9B, all yeast transformants grew well on SD/–Trp medium. However, only yeast transformants containing pBD-SlSR1 or pBD-SlSR3L were able to grow on the SD/–Trp/–His medium and produced a blue pigment after the addition of x-?-gal, showing a ?-galactosidase activity, whereas transformants containing the pBD empty vector did not. These results indicate that both SlSR1 and SlSR3L have transactivation activity in yeasts. Taken together, our experimental data demonstrate that both SlSR1 and SlSR3L are nucleus-localized transcriptional activators.
Subcellular localization and transactivation activity of SlSR1 and SlSR3L proteins. (A)SlSR1 and SlSR3L are localized in nucleus. Agrobacteria carrying pFGC-Egfp-SlSR1, pFGC-Egfp-SlSR3L or pFGC-Egfp were infiltrated intoN. benthamianaleaves and the images were taken in dark field for green fluorescence (left), in white field for the morphology of the cell (middle), and in combination (right), respectively.(B)SlSR1 and SlSR3L have transactivation activity. Yeast cells carrying pBD-SlSR1, pBD-SlSR3L or pBD empty vector (as a negative control) were streaked on SD/–Trp plates (left) or SD/–Trp/–His plates (middle) for 3 days at 28°C. The x-?-gal was added to the SD/-Trp/-His plates and kept at 28°C for 6 hr (right).
Discussion
Previous studies have identified a total of sevenSlSRsgenes in tomato and found that expression ofSlSRswas regulated in tomato fruit by developmental cues and by biotic and abiotic environmental stress signals [29],[30.]; however, the biological functions ofSlSRsin tomato response to biotic and abiotic stress remain unclear. In the present study, data from our VIGS-based functional analyses demonstrate that both SlSR1 and SlSR3L act as negative regulators of defense response againstB. cinereaandPstDC3000虽然SlSR1L函数作为一个积极的边条tor of drought stress tolerance in tomato. These findings not only demonstrate that members of the small SlSR family play important roles in regulation of defense responses to biotic and abiotic stresses but also extend our understanding on the biological function ofSlSRsin regulating stress response.
In plants, members of the SR family have been shown to be induced by infections from different pathogens. For example, the expression ofAtSR1in Arabidopsis was induced byPstDC3000 andG. cichoracearum[21]. In this study, we observed that the expression ofSlSRscould be induced with different patterns byPstDC3000 andB. cinerea(Figures1and2). In response to infection ofB. cinerea, all theSlSRsshowed upregulated expression patterns in leaf tissues, especially forSlSR1,SlSR1L,SlSR3LandSlSR4, whose expression levels were significantly upregulated (Figure1). TheB. cinerea-induced expression patterns ofSlSRsin leaf tissues are somewhat different from the previous observation that the expressions ofSlSR1,SlSR1L,SlSR2LandSlSR3in wounded tomato fruit were suppressed byB. cinerea[30.], probably due to the tissue-specific expression feature of theSlSRs[29]. In contrast to theB. cinerea-induced expression patterns ofSlSRs, expression ofSlSR1,SlSR1L,SlSR3andSlSR3Lin leaf tissues was upregulated in response to infection ofPstDC3000 (Figure2). ThePstDC3000-induced expression ofSlSR1类似于阿f the ArabidopsisAtSR1, closely related toSlSR1[29], whose expression was induced byPstDC3000 [21]. However, infection ofPstDC3000 did not affect the expression ofSlSR2andSlSR3but suppressed the expression ofSlSR4in leaf tissues (Figure2), which is contrast to the observation that the expression ofSlSR4in fruit tissues was induced byB. cinerea[30.]. Therefore, it seems reasonable that the expression ofSlSRsis precisely controlled by complex mechanisms in response to infection from different pathogens. On the other hand, theB. cinerea- andPstDC3000-induced expressions ofSlSRs, especially forSlSR1andSlSR3L, were much evident after 24 hr of inoculation (Figures1and2), which is similar to the observation that the expression ofAtSR1in Arabidopsis leaves was only significantly induced byPstDC3000 after 24 hr of inoculation [21]. It was also found that the expression of mostSlSRsin tomato fruits was markedly induced by exogenous SA and MeJA only after 8 hr of treatment [30.]. These observations suggest that most of theSlSRsshould belong to late pathogen-responsive genes [30.]. TheB. cinerea- andPstDC3000-induced expression ofSlSR1andSlSR3Lin leaves (Figures1and2), along with their SA- and MeJA-induced expression patterns in fruits [30.], suggest that SlSR1 and SlSR3L may play important roles in defense response to infection ofB. cinereaandPstDC3000.
In our VIGS-based functional analyses ofSlSRsin defense response against different pathogens, we found that silencing of eitherSlSR1orSlSR3Lresulted in increased disease resistance againstB. cinereaandPstDC3000, as theSlSR1- andSlSR3L-silenced plants exhibited less severity of the diseases and supported lessin plantagrowth of the pathogens than the non-silenced plants (Figures4and5), indicating that loss-of-function of eitherSlSR1orSlSR3Lconfers a broad-spectrum disease resistance against different pathogens with distinct infection styles. SlSR1 is phylogenetically closely related to the Arabidopsis AtSR1 [29], whose loss-of-function mutant plants showed increased disease resistance against three different pathogens includingPstDC3000,B. cinereaandG. cichoracearum[19]-[21] and gain-of-function mutant plants exhibited compromised systemic acquired resistance and basal immunity [21],[24]. Similar results were also observed in the mutant plants of theOsCBT基因水稻SR家庭成员密切相关AtSR1andSlSR1, which exhibited significant enhanced resistance to blast fungal pathogenM. griseaand leaf blight bacterial pathogenX. oryzaepv.oryzae[17]. Thus, it is likely that loss-of-function of some of the SR family members can confer broad-spectrum disease resistance in plants. This may have extensive significance that members of the SR family can be used for generating transgenic varieties with broad-spectrum resistance in economically important crops through RNA interference-mediated suppression of expression of target SR genes. Notably, in addition to SR1, the function of other SR family members in disease resistance has not been elucidated so far. In this study, we demonstrated that SlSR3L, like SlSR1 in tomato (Figures4and5) and AtSR1 in Arabidopsis [19]-[21], acts as a negative regulator of disease resistance against different pathogens in tomato. This finding characterized one member with important function in disease resistance from the relatively small SR family. It will be interesting to examine whether the Arabidopsis homologs of SlSR3L, AtSR3 and AtSR6 [29], play roles in regulating disease resistance. Although expression ofSlSR1L,SlSR2,SlSR2LandSlSR3was induced byB. cinereaand/orPstDC3000 (Figures1and2),每一种的沉默SlSRgenes had no effect on disease resistance against these two pathogens (Figures4and5). The expression ofSlSR4was induced byB. cinereabut suppressed byPstDC3000 (Figures1and2); however, silencing ofSlSR4did not lead to any alteration in disease resistance toB. cinereaandPstDC3000 (Figures4and5). These results indicate that theSlSRgenes are highly responsive to pathogen infection but the responsiveness of theseSlSRgenes may be a side effect caused by pathogen infection but not a true reflection for their function in disease resistance.
The observed increased disease resistance againstB. cinereaandPstDC3000 in theSlSR1- andSlSR3L-silenced plants may be attributed to an improved basic immunity resulted from loss-of-function ofSlSR1andSlSR3L.This hypothesis is supported by several lines of evidence obtained from some biochemical and molecular analyses toward theSlSR1- andSlSR3L-silenced plants under pathogen-free conditions. Firstly, theSlSR1- andSlSR3L-silenced plants constitutively accumulated high level of H2O2in leaves, as revealed byin situDAB staining (Figure6A). Similar phenomenon was also observed in the Arabidopsissr1mutant plants that accumulated high level of H2O2in leaves without pathogen infection [19],[20]. It was previously found that suppression ofSlWfi1expression resulted in significant decrease of H2O2accumulation in antisense tomato plants [46]. The expression ofSlWfi1andSlRboh1, encoding plasma membrane-localized NADPH oxidases that are involved in generating ROS [33], was significantly upregulated in theSlSR1- andSlSR3L-silenced plants (Figure6B), indicating an accelerated generation of ROS in these plants. By contrast, the ROS scavenging capacity in theSlSR1- andSlSR3L-silenced plants might not be affected significantly as the expression of theSODgene was not changed (Figure6B). The upregulated expression of theCATandAPXgenes in theSlSR1- andSlSR3L-silenced plants might be a response to the high level of H2O2accumulated in these plants as H2O2was found to be capable of mediating the expression ofCATgenes under stress conditions [47],[48]. An accelerated generation with an unchanged scavenging capacity in theSlSR1- andSlSR3L-silenced plants broke the ROS generating and scavenging balance and favored to accumulate high level of H2O2, which in turn acts as signaling molecules to activate defense response against pathogens. Secondly, theSlSR1- andSlSR3L-silenced plants constitutively expressed defense genes and PTI marker genes. Similar to the observations that the Arabidopsissr1和大米oscbtmutant plants constitutively expressed a diverse set of defense genes [17],[19],[20], constitutive high levels of expression of defense genes includingSlPR1a,SlPR1b,SlPR-P2,SlPIN2andSlLapA, which are regulated by different signaling pathways, were observed in theSlSR1- andSlSR3L-silenced plants (Figure6B). In addition, the expression of the PTI marker genes includingPti5,Lrr22andWRKY28[34],[35] was also significantly upregulated in theSlSR1- andSlSR3L-silenced plants (Figure6C). These data indicate that both SlSR1 and SlSR3L negatively regulate PTI response. In this regarding, silencing of eitherSlSR1orSlSR3Lwould relieve their suppression on PTI response and thus lead to increased resistance against multiple pathogens includingB. cinereaandPstDC3000 (Figures4and5). This is supported by general knowledge that PTI but not effector-triggered immunity (ETI) plays important roles in regulating immunity against necrotrophic fungal pathogens likeB. cinerea[49] while both the PTI and ETI are require for immunity to biotrphic/hemibiotrophic pathogens such asPstDC3000 [50]. Thirdly, theSlSR1- andSlSR3L-silenced plants constitutively activated the SA- and ET-mediated defense signaling pathways. It was previously found that the Arabidopsissr1mutant plants contained high level of SA and had upregulated expression of defense and signaling genes [19],[20] and that AtSR1 can bind to the CGCG box in the promoters ofEDS1,NDR1andEIN3[20],[21], indicating the involvements of AtSR1 in the SA- and ET-mediated signaling pathways. Our qRT-PCR analyses of expression of some defense signaling pathway-associated genes also demonstrated that both SlSR1 and SlSR3L have functions that negatively regulate the SA- and ET-mediated signaling pathways in tomato, as revealed by the upregulated expression of theSlNPR1,SlEDS1,SlTGA1,SlETR4andSlERF1genes in theSlSR1- andSlSR3L-silenced plants (Figure6C). This is partially supported by the facts that the expression ofSlSR1andSlSR3Lin tomato fruits was induced rapidly by exogenously applied SA and ET [29],[30.]. In particular, it was shown that the Arabidopsis AtSR1 could bind to the CGCG box, a characteristiccis-elements for the SR proteins [12], in the promoter region ofEDS1and suppressed the expression ofEDS1[20], which is critical to biosynthesis of SA. Bioinformatics analysis also identified a CGCG box within 1.3 Kb from the starting codon in the promoter of tomatoSlEDS1gene [30.]. It is thus possible that SlSR1 and perhaps SlSR3Ll regulate negatively the expression ofSlEDS1through binding to the CGCG box as the expression level ofSlEDS1was upregulated when eitherSlSR1orSlSR3Lwas silenced (Figure6C). Furthermore, the expression ofSlJAZ1, known to be associated with the JA-mediated signaling pathway [39], was downregulated in theSlSR1- andSlSR3L-silenced plants (Figure6C), suggesting a negative impact of SlSR1 and SlSR3L on the JA-mediated signaling pathway. This is in agreement with the observations that the Arabidopsis AtSR1 is a negative regulator for JA biosynthesis and herbivory tolerance [22],[23]. Another possibility that the constitutively activated SA-mediated signaling pathway in theSlSR1- andSlSR3L-silenced plants antagonistically suppressed the JA-mediated signaling pathway cannot be ruled out because the antagonistic cross-talk between these two signaling pathways is a common phenomenon occurred in regulating defense response against infection by different pathogens [51]. Taken together, these data suggest that both SlSR1 and SlSR3L negatively regulate basic immunity in tomato through modulating the SA- and ET-mediated signaling pathways.
The involvement of the SR family members in abiotic stress was recently investigated using Arabidopsis knockout mutant lines [25],[27]. Our VIGS-based functional analyses discovered that silencing ofSlSR1Lresulted in decreased drought stress tolerance (Figures7E and8A). SlSR1L is phylogenetically related to the Arabidopsis AtSR2 [29], which was shown to regulate drought stress responses [27]. Thus, it is likely that SlSR1L plays an important role in regulation of drought stress tolerance in tomato. The expression ofSlSR1Lwas induced significantly by drought stress in detached leaves and in whole plants (Figure7A and D). The decreased drought tolerance in theSlSR1L-silenced plants might be caused by multiple factors including morphological, physiological and molecular changes, which are affected by loss-of-function ofSlSR1L.Like the stunted primary root in the Arabidopsissr2mutant plants [27], theSlSR1L-silenced plants had limited root system and biomass under normal growth condition (Figure8C and D). Although the exact function ofSlSR1Lin development of the root system in tomato needs to be further investigated, the limited root system in theSlSR1L-silenced plants may result in reduced capacity of water uptake. Another fact that affected the water status in theSlSR1L-silenced plants was the accelerated rate of water loss, as revealed in the detached leaves (Figure8B). Furthermore, the expression ofSlAREB1[42],SlDREB[43],SlSpUSP[44],SlAREB2[42] andSGN-213276[40] was suppressed in theSlSR1L-silenced plants (Figure8E), indicating that SlSR1L may regulate the expression of a large set of drought stress-responsive genes. Recent microarray-based analyses of gene expression profiling between the Arabidopsissr1and wild type plants revealed that hormone-mediated signaling such as ABA-mediated signaling plays important roles in AtSR1-regulated abiotic stress response [18],[27]. However, exogenous ABA did not induce the expression ofSlSR1Lin tomato (Figure7B) andAtSR2in Arabidopsis [12]. Thus, it is likely that, as a transcription factor, SlSR1L acts in a yet-unknown signaling pathway, in which some hormones such as ABA are involved, to regulate drought stress response in tomato.
我们的数据提出了研究清楚地示威te that SlSR1/SlSR3L and SlSR1L play important roles in biotic and abiotic stress responses, respectively. However, several questions regarding the mechanism of action of SlSR1/SlSR3L and SlSR1L in biotic and abiotic stress response need to be addressed. Further identification of downstream target genes regulated by SlSR1/SlSR3L and SlSR1L will help to elucidate the molecular mechanisms and the signaling pathways involved in the SlSR1/SlSR3L-regulated defense response against pathogens and the SlSR1L-regulated drought stress response. Of particular, the SlSR3L will be a priority for further study because the function of its orthologs in other plants such as the Arabidopsis AtSR3 and AtSR6 [29] has not been defined yet. On the other hand, biochemical studies have shown that the Arabidopsis AtSR2/CAMTA1 [11], rice OsCBT [14] and tomato SlSR1 and SlSR3L (Figure9) are functionally transcriptional activators in yeast. However, the Arabidopsis AtSR1 was shown to bind to the CGCG box in the promoters of theEDS1,NDR1andEIN3genes and repress their expression [20],[21]. Thus, the biochemical mechanism regarding how the SR proteins as transcriptional activators repress the expression of the target genes after binding to the CGCG box in the promoters of these genes needs to be further investigated.
Conclusion
Tomato genome encodes sevenSlSRgenes and expression ofSlSR1andSlSR3Lwas significantly induced byB. cinereaandPstDC3000. Silencing of eitherSlSR1orSlSR3Lresulted in enhanced resistance toB. cinereaandPstDC3000 and led to constitutive accumulation of H2O2, elevated expression of defense genes, PTI marker genes and regulatory genes involved in the SA- and ET-mediated signaling pathways. Meanwhile, expression ofSlSR1Lwas significantly induced by drought stress and silencing ofSlSR1Lled to decreased drought stress tolerance. These results demonstrate that both SlSR1 and SlSR3L in the tomatoSlSR/CAMTAfamily are negative regulators of defense response againstB. cinereaandPstDC3000 while SlSR1L is a positive regulator of drought stress tolerance in tomato.
Methods
Plant growth and treatments
Tomato (Solanum lycopersicum) cv. Suhong 2003 was used for all experiments. Seeds were scarified on moist filter paper in Petri dishes for 2 days and the sprouted seeds were transferred into a mixture of perlite: vermiculite: plant ash (1:6:2). All tomato plants were grown in a growth room at 22–24°C with 60% relative humidity (RH) under a 14 hr light (350 μmol · s-1· m-2photons m-2sec-1)/10 hr dark cycle. Two-week-old plants were used for VIGS assays and four-week-old plants were used for other experiments. For analysis of gene expression in response to pathogen infection, whole plant inoculation assays forB. cinereaand vacuum infiltrated inoculation assays forPstDC3000连同相应mock-inoculation controls were performed (see below). For analysis of gene expression in drought stress, drought stress was applied to the plants by stopping watering for a period until wilting symptom appeared and normally watered plants were used as controls in the whole plant drought stress assays. Alternatively, fully expanded leaves were detached and subjected to drought stress treatment by placing on lab blench or on water-saturated filter papers in Petri dishes as controls in detached leaf assays. For ABA treatment, a solution of ABA (100 ?M) or same volume of water as a control was sprayed onto leaf surface of the tomato plants. Leaf samples were collected at indicated time points after treatment or inoculation and stored at -80°C until use.
Plant inoculation and disease assays
PstDC3000 was grown overnight in King’s B (KB) liquid medium containing rifampicin at 50 ?g/ml. The bacteria were collected and resuspended in 10 mM MgCl2to OD600?=?0.0002 for plant inoculation. Four-week-old plants were vacuum infiltrated with suspension ofPstDC3000 or with MgCl2solution as a mock inoculation control. The inoculated plants were kept in a sealed container to maintain high humidity (RH?>?90%) and disease progress was observed daily. Leaf samples were collected from at least sixPstDC3000-inoculated or mock-inoculated plants at different time points after inoculation and used for analysis of gene expression andin plantabacterial growth. For measurement of bacterial growth, leaf discs (6 mm in diameter) were surface sterilized in 70% ethanol for 10 s, homogenized in 200 ?l of 10 mM MgCl2, diluted in 10 mM MgCl2, and plated on KB agar plates containing rifampicin at 50 ?g/ml. The plates were incubated at 28°C and the bacterial numbers were counted 3 days after incubation.
B. cinereawas grown on 2× V8 agar (36% V8 juice, 0.2% CaCO3, and 2% agar) at 22°C and spores were collected and resuspended in 1% maltose buffer to 2 × 105spores/ml for whole plant inoculation and 1 × 105spores/ml for detached leaf inoculation. Whole plant inoculation and detached leaf inoculation assays were performed according to previously reported procedure [52],[53]. In the detached leaf inoculation assays, leaves were detached from at least 10 four-week-old VIGS-infiltrated plants and inoculated by dropping 5 ?l of spore suspension on leaf surface. In the whole plant inoculation assays, four-week-old plants were inoculated by foliar spraying with spore suspension ofB. cinereaor with same volume of 1% maltose buffer as a mock-inoculation control and leaf samples were collected at different time points after inoculation for analysis of gene expression andin plantafungal growth. The inoculated leaves and plants were kept at 22°C in sealed containers to retain the moist conditions favorable for disease development.In plantafungal growth was analyzed by the amplification of the transcripts of aB. cinereaActin gene as a marker [53],[54] using a pair of primersBcActin-F andBcActin-R (Additional file2). Relative fungal growth was expressed as folds of the transcript levels ofBcActinvs the transcript levels of a tomatoActingene.
Construction of vectors and VIGS assays
Based on the cDNA sequences of all sevenSlSRgenes [29], fragments of 386–462 bp for eachSlSRgene (Additional file3) were amplified with gene-specific primers (Additional file2) from cDNAs synthesized from total RNA prepared from tomato leaf samples. After cloning and sequencing, these VIGS fragments were cloned into pTRV2 vector [32),收益率pTRV2-SlSRs。这些构造pTRV2-SlSRts were then introduced intoAgrobacterium tumefaciensstrain GV3101 by electroporation using GENE PULSER II Electroporation System (Bio-Rad Laboratories, Hercules, CA, USA). Agrobacteria carrying pTRV2-GUS (as a negative control), pTRV2-PDS (as a positive control for silencing efficiency examination) or pTRV2-SlSRs were cultivated in YEP medium (10 g/l peptone, 10 g/l yeast extract, 5 g NaCl/l, 50 ?g/ml rifampicin, 50 ?g/ml kanamycin and 25 ?g/ml gentamicin) for 36 hr with continuous shaking in a 28°C incubator. Cells were centrifuged and resuspended in infiltration buffer (10 mM MgCl2, 150 ?M acetosyringone, MES, pH5.7). The agrobacteria carrying pTRV2-GUS, pTRV2-PDS or pTRV2-SlSR were mixed with the agrobacteria carrying pTRV1 in a ratio of 1:1 and maintained at OD6001.5 = ? 3小时在室温下。混合agrobacteria suspension was infiltrated into the abaxial surface of the 2-week-old seedlings using 1 mL needleless syringes. Efficiency of the silencing procedure was evaluated based on the appearance of bleaching phenotype in the pTRV2-PDS-infiltrated plants [32]. When more than 90% of the pTRV2-PDS-infiltrated plants showed bleaching phenotype, the pTRV2-GUS- or pTRV2-SlSR-infiltrated plants in an independent experiment with same VIGS procedure were used for further study. Leaf samples were collected at 2 weeks after VIGS infiltration and used for analysis of the silencing efficiency by qRT-PCR.
Subcellular localization
The coding sequences ofSlSR1andSlSR3Lwere PCR amplified using pairs of gene-specific primers SlSR1-3F/SlSR1-3R and SlSR3L-3F/SlSR3L-3R, respectively (Additional file2). After confirmation by sequencing, the coding regions of theSlSR1andSlSR3Lgenes were cloned into pFGC-Egfp. The recombinant plasmids pFGC-Egfp-SlSR1, pFGC-Egfp-SlSR3L and pFGC-Egfp were transformed intoA. tumefaciesstrain GV3101 and the transformed agrobacteria were infiltrated individually into leaves of four-week-oldN. benthamianaplants using 1-ml needless syringes. These agroinfiltrated plants were allowed to grow in a growth chamber at 25°C for 48 hr, and the GFP fluorescence was examined under a Leica TCS SP5 laser confocal microscope with excitation wavelength of 488 nm.
Transcription activation assay in yeast
The coding sequences ofSlSR1andSlSR3Lwere amplified using gene-specific primers (Additional file2) and cloned into pBD-GAL4Cam vector to yield pBD-SlSR1 and pBD-SlSR3L. These two recombinant plasmids and the pBD empty vector (as a negative control) were transformed into yeast strain AH109. The transformed yeasts were confirmed by colony PCR and then cultivated on the SD/–Trp and SD/–Trp/–His medium for 3 days at 28°C, followed by addition of x-?-gal (5-bromo-4-chloro-3-indolyl-?-D-galactopyranoside). Transactivation activity of the fused proteins was evaluated according to the growth situation and production of blue pigments after the addition of x-?-gal of the transformed yeast cells on the SD/–Trp/–His medium.
Detection of H2O2 accumulation
Detection ofin situH2O2was carried out using DAB staining method as described previously [55]. Leaves were collected from pTRV2-SlSR1- or pTRV2-SlSR3L-infiltrated and pTRV2-GUS-infiltrated plants and dipped into DAB solution (1 mg/ml, pH3.8) and incubated for 8 hr in dark at room temperature. Thereafter, leaves were placed into acetic acid/glycerol/ethanol (1:1:1, vol/vol/vol) and boiled for 5 min in a water bath. After several changes of the solution, then leaves were maintained in 60% glycerol and accumulation of H2O2in leaves was photographed using a digital camera.
Drought tolerance assays
At least 10 individual pTRV2-SlSRs- or pTRV2-GUS-infiltrated plants were used in each experiment. Drought stress was applied to the tomato plants by stopping watering for a certain period of 10–12 days or until the wilting symptoms were obvious. To assess the ratio of water loss, fully expanded leaves from 6 individual pTRV2-SlSR1L- or pTRV2-GUS-infiltrated plants were detached and placed on the bench top. The weights of the leaves were recorded at different time points after detachment and the average water loss ratio was calculated by comparing with the initial weights. Roots from 6 individual pTRV2-SlSR1L- or pTRV2-GUS-infiltrated plants were cut, cleaned and dried in 70°C oven for 24 hr and the final weight was calculated and compared. Leaf samples were collected from the pTRV2-SlSRs- or pTRV2-GUS-infiltrated plants at 0 hr (as unstressed control) and 7 days after stopping watering and were subjected to analysis of gene expression.
qRT-PCR analysis of gene expression
Total RNA was extracted from frozen leaf samples using TRIzol reagent (Invitrogen, Shanghai, China) and treated with RNase-free DNase (TaKaRa, Dalian, China) to erase any genomic DNA in the RNA samples. First-strand cDNA was synthesized from 0.6 ?g total RNA using AMV reverse transcriptase (TaKaRa, Dalian, China) according to the manufacturer’s recommendations. Each qRT-PCR reaction contained 12.5 ?l SYBR Premix Ex Taq (TaKaRa, Dalian, China), 0.1 ?g cDNA and 7.5 pmol of each gene-specific primer (Additional file2) in a final volume of 25 ?l, and run on three independent biological replicates. The qRT-PCR was performed in a CFX96 real-time PCR detection system (BioRad, Hercules, CA, USA) and relative expression levels were calculated using the 2-??CTmethod. The expression level of a tomato actin gene was used as an internal control to normalize the expression data for the target genes. Relative expression levels of the target genes were shown as folds of the expression level of the actin gene or as folds of the expression levels in treated plants/control plants.
Statistical analysis
All experiments were repeated independently three times and data were collected from experiments on three biological samples. All data obtained were subjected to statistical analysis according to the Student’st-test and the probability values ofp?
Accession numbers forSlSRs
The tomatoSlSRgene sequences used in this study were retrieved from GenBank under the following accession numbers:SlSR1, GU170838;SlSR1L, JN558810;SlSR2, JN566047;SlSR2L, JN566048;SlSR3, JN566049;SlSR3L, JN566051;SlSR4, JN566050. TheseSlSRgene sequences were deposited by Yang et al. [29].
Availability of supporting data
The sequences of SlSR proteins and sequences of VIGS fragments forSlSRgenes used in this study are included in Additional files1and3.
Authors’ contributions
XL, LH, YZ, ZO, YH, HZ and DL carried out the experiments. XL and FS designed the experiments. FS and XL wrote the paper. All authors read and approved the final manuscript.
Additional files
References
- 1.
Kudla J, Batistic O, Hashimoto K: Calcium signals: the lead currency of plant information processing. Plant Cell. 2010, 22 (3): 541-563. 10.1105/tpc.109.072686.
- 2.
McAinsh MR, Pittman JK: Shaping the calcium signature. New Phytol. 2009, 181 (2): 275-294. 10.1111/j.1469-8137.2008.02682.x.
- 3.
Plieth C: Signal percolation through plants and the shape of the calcium signature. Plant Signal Behav. 2010, 5 (4): 379-385. 10.4161/psb.5.4.10717.
- 4.
Ma W, Berkowitz GA: The grateful dead: calcium and cell death in plant innate immunity. Cell Microbiol. 2007, 9 (11): 2571-2585. 10.1111/j.1462-5822.2007.01031.x.
- 5.
Reddy ASN, Ali GS, Celesnik H, Day IS: Coping with stresses: roles of calcium- and calcium/calmodulin-regulated gene expression. Plant Cell. 2011, 23 (6): 2010-2032. 10.1105/tpc.111.084988.
- 6.
Sarwat M, Ahmad P, Nabi G, Hu X: Ca2+signals: the versatile decoders of environmental cues. Crit Rev Biotechnol. 2013, 33 (1): 97-109. 10.3109/07388551.2012.672398.
- 7.
Poovaiah BW, Du LQ, Wang HZ, Yang TB: Recent advances in calcium/calmodulin-mediated signaling with an emphasis on plant-microbe interactions. Plant Physiol. 2013, 163 (2): 531-542. 10.1104/pp.113.220780.
- 8.
Zielinski RE: Calmodulin and calmodulin-binding proteins in plants. Annu Rev Plant Biol. 1998, 49: 697-725. 10.1146/annurev.arplant.49.1.697.
- 9.
Bouche N, Yellin A, Snedden WA, Fromm H: Plant-specific calmodulin-binding proteins. Annu Rev Plant Biol. 2005, 56: 435-466. 10.1146/annurev.arplant.56.032604.144224.
- 10.
Luan S, Kudla J, Rodriguez-Concepcion M, Yalovsky S, Gruissem W: Calmodulins and calcineurin Bac like proteins: calcium sensors for specific signal response coupling in plants. Plant Cell. 2002, 14 (Suppl): S389-S400.
- 11.
Bouche N, Scharlat A, Snedden W, Bouchez D, Fromm H: A novel family of calmodulin-binding transcription activators in multicellular organisms. J Biol Chem. 2002, 277 (24): 21851-21861. 10.1074/jbc.M200268200.
- 12.
Yang TB, Poovaiah BW: A calmodulin-binding/CGCG box DNA-binding protein family involved in multiple signaling pathways in plants. J Biol Chem. 2002, 277 (47): 45049-45058. 10.1074/jbc.M207941200.
- 13.
Finkler A, Ashery-Padan R, Fromm H: CAMTAs: calmodulin-binding transcription activators from plants to human. FEBS Lett. 2007, 581 (21): 3893-3898. 10.1016/j.febslet.2007.07.051.
- 14.
Choi MS, Kim MC, Yoo JH, Moon BC, Koo SC, Park BO, Lee JH, Koo YD, Han HJ, Lee SY, Chung WS, Lim CO, Cho MJ: Isolation of a calmodulin-binding transcription factor from rice (Oryza sativaL.). J Biol Chem. 2005, 280 (49): 40820-40831. 10.1074/jbc.M504616200.
- 15.
Reddy ASN, Reddy VS, Golovkin M: A calmodulin binding protein from Arabidopsis is induced by ethylene and contains a DNA-binding motif. Biochem Biophys Res Commun. 2000, 279 (3): 762-769. 10.1006/bbrc.2000.4032.
- 16.
Yang TB, Poovaiah BW: An early ethylene up-regulated gene encoding a calmodulin-binding protein involved in plant senescence and death. J Biol Chem. 2000, 275 (49): 38467-38473. 10.1074/jbc.M003566200.
- 17.
Koo SC, Choi MS, Chun HJ, Shin DB, Park BS, Kim YH, Park HM, Seo HS, Song JT, Kang KY, Yun DJ, Chung WS, Cho MJ, Kim MC: The calmodulin-binding transcription factor OsCBT suppresses defense responses to pathogens in rice. Mol Cells. 2009, 27 (5): 563-570. 10.1007/s10059-009-0081-4.
- 18.
Galon Y, Aloni R, Nachmias D, Snir O, Feldmesser E, Scrase-Field S, Boyce JM, Bouche N, Knight MR, Fromm H: Calmodulin-binding transcription activator 1 mediates auxin signaling and responds to stresses in Arabidopsis. Planta. 2010, 232 (1): 165-178. 10.1007/s00425-010-1153-6.
- 19.
Galon Y, Nave R, Boyce JM, Nachmias D, Knight MR, Fromm H: Calmodulin-binding transcription activator (CAMTA) 3 mediates biotic defense responses in Arabidopsis. FEBS Lett. 2008, 582 (6): 943-948. 10.1016/j.febslet.2008.02.037.
- 20.
Du LQ, Ali GS, Simons KA, Hou JG, Yang TB, Reddy ASN, Poovaiah BW: Ca2+/calmodulin regulates salicylic-acid-mediated plant immunity. Nature. 2009, 457 (7233): 1154-1158. 10.1038/nature07612.
- 21.
Nie HZ, Zhao CZ, Wu GH, Wu YY, Chen YF, Tang DZ: SR1, a calmodulin-binding transcription factor, modulates plant defense and ethylene-induced senescence by directly regulating NDR1 and EIN3. Plant Physiol. 2012, 158 (4): 1847-1859. 10.1104/pp.111.192310.
- 22.
Laluk K, Prasad KVSK, Savchenko T, Celesnik H, Dehesh K, Levy M, Mitchell-Olds T, Reddy ASN: The calmodulin-binding transcription factor signal responsive1 is a novel regulator of glucosinolate metabolism and herbivory tolerance in Arabidopsis. Plant Cell Physiol. 2012, 53 (12): 2008-2015. 10.1093/pcp/pcs143.
- 23.
Qiu Y, Xi J, Du L, Suttle JC, Poovaiah BW: Coupling calcium/calmodulin-mediated signaling and herbivore-induced plant response through calmodulin-binding transcription factor AtSR1/CAMTA3. Plant Mol Biol. 2012, 79 (1–2): 89-99. 10.1007/s11103-012-9896-z.
- 24.
Jing B, Xu S, Xu M, Li Y, Li S, Ding J, Zhang Y: Brush and spray: a high throughput systemic acquired resistance assay suitable for large scale genetic screening. Plant Physiol. 2011, 157 (3): 973-980. 10.1104/pp.111.182089.
- 25.
Doherty CJ, Buskirk HAV, Myers SJ, Thomashow MF: Roles for Arabidopsis CAMTA transcription factors in cold-regulated gene expression and freezing tolerance. Plant Cell. 2009, 21 (3): 972-984. 10.1105/tpc.108.063958.
- 26.
Kim YS, Park SC, Gilmour SJ, Thomashow MF: Roles of CAMTA transcription factors and salicylic acid in configuring the low-temperature transcriptome and freezing tolerance of Arabidopsis. Plant J. 2013, 75 (3): 364-376. 10.1111/tpj.12205.
- 27.
Pandey N, Ranjan A, Pant P, Tripathi RK, Ateek F, Pandey HP, Patre UV, Sawant SV: CAMTA1 regulates drought responses inArabidopsis thaliana.BMC Genomics. 2013, 14: 216-10.1186/1471-2164-14-216.
- 28.
Zegzouti H, Jones B, Frasse P, Marty C, Maitre B, Latche AA, Pech JC, Bouzayen M: Ethylene-regulated gene expression in tomato fruit: characterization of novel ethylene-responsive and ripening-related genes isolated by differential display. Plant J. 1999, 18 (6): 589-600. 10.1046/j.1365-313x.1999.00483.x.
- 29.
Yang TB, Peng H, Whitaker BD, Conway WS: Characterization of a calcium/calmodulin regulated SR/CAMTA gene family during tomato fruit development and ripening. BMC Plant Biol. 2012, 12: 19-10.1186/1471-2229-12-19.
- 30..
Yang TB, Peng H, Whitaker BD, Jurick WM: Differential expression of calcium/calmodulin-regulatedSlSRsin response to abiotic and biotic stresses in tomato fruit. Physiol Plant. 2013, 148 (3): 445-455. 10.1111/ppl.12027.
- 31.
Kawazu K, Mochizuki A, Sato Y, Sugeno W, Murata M, Seo S, Mitsuhara I: Different expression profiles of jasmonic acid and salicylic acid inducible genes in the tomato plant against herbivores with various feeding modes. Arthropod Plant Interact. 2012, 6: 221-230. 10.1007/s11829-011-9174-z.
- 32.
Liu Y, Schiff M, Dinesh-Kumar SP: Virus-induced gene silencing in tomato. Plant J. 2002, 31 (6): 777-786. 10.1046/j.1365-313X.2002.01394.x.
- 33.
Marino D, Dunand C, Puppo A, Pauly N: A burst of plant NADPH oxidases. Trends Plant Sci. 2012, 17 (1): 9-15. 10.1016/j.tplants.2011.10.001.
- 34.
Kim JG, Li X, Roden JA, Taylor KW, Aakre CD, Su B, Lalonde S, Kirik A, Chen Y, Baranage G, McLane H, Martin GB, Mudgett MB:XanthomonasT3S effector XopN suppresses PAMP-triggered immunity and interacts with a tomato atypical receptor-like kinase and TFT1. Plant Cell. 2009, 21 (4): 1305-1323. 10.1105/tpc.108.063123.
- 35.
Nguyen HP, Chakravarthy S, Velásquez AC, McLane HL, Zeng L, Nakayashiki H, Park DH, Collmer A, Martin GB: Methods to study PAMP-triggered immunity using tomato andNicotiana benthamiana.Mol Plant-Microbe Interact. 2010, 23 (8): 991-999. 10.1094/MPMI-23-8-0991.
- 36.
Ekengren SK, Liu Y, Schiff M, Dinesh-Kumar SP, Martin GB: Two MAPK cascades, NPR1, and TGA transcription factors play a role inPto-mediated disease resistance in tomato. Plant J. 2003, 36 (6): 905-917. 10.1046/j.1365-313X.2003.01944.x.
- 37.
Kamiyoshihara Y, Tieman DM, Huber DJ, Klee HJ: Ligand-induced alterations in the phosphorylation state of ethylene receptors in tomato fruit. Plant Physiol. 2012, 160 (1): 488-497. 10.1104/pp.112.202820.
- 38.
Di Matteo A, Ruggieri V, Sacco A, Rigana MM, Carriero F, Bolger A, Fernie AR, Frusciante L, Barone A: Identification of candidate genes for phenolics accumulation in tomato fruit. Plant Sci. 2013, 205–206: 87-96. 10.1016/j.plantsci.2013.02.001.
- 39.
Pauwels L, Goossens A: The JAZ proteins: a crucial interface in the jasmonate signaling cascade. Plant Cell. 2011, 23 (9): 3089-3100. 10.1105/tpc.111.089300.
- 40.
Gong PJ, Zhang J, Li H, Yang C, Zhang C, Zhang X, Khurram Z, Zhang Y, Wang T, Fei Z, Ye Z: Transcriptional profiles of drought-responsive genes in modulating transcription signal transduction, and biochemical pathways in tomato. J Exp Bot. 2010, 61 (13): 3563-3575. 10.1093/jxb/erq167.
- 41.
Wasilewska A, Vlad F, Sirichandra C, Redko Y, Jammes F, Valon C, Freid it Frey N, Leung J: An update on abscisic acid signaling in plants and more….. Mol Plant. 2008, 1 (2): 198-217. 10.1093/mp/ssm022.
- 42.
Orellana S, Yanez M, Espinoza A, Verdugo I, Gonzalez E, RuizLara S, Casaretto JA: The transcription factor SlAREB1 confers drought, salt stress tolerance and regulates biotic and abiotic stress-related genes in tomato. Plant Cell Environ. 2010, 33 (12): 2191-2208. 10.1111/j.1365-3040.2010.02220.x.
- 43.
Li J, Sima W, Ouyang B, Wang TT, Ziaf K, Luo ZD, Liu LF, Li HX, Chen ML, Huang YQ, Feng YQ, Hao YH, Ye ZB: TomatoSlDREBgene restricts leaf expansion and internode elongation by downregulating key genes for gibberellin biosynthesis. J Exp Bot. 2012, 63 (18): 6407-6420. 10.1093/jxb/ers295.
- 44.
Loukehaich R, Wang T, Ouyang B, Ziaf K, Li H, Zhang J, Lu Y, Ye Z: SpUSP, an annexin-interacting universal stress protein, enhances drought tolerance in tomato. J Exp Bot. 2012, 63 (15): 5593-5606. 10.1093/jxb/ers220.
- 45.
Guo Y, Huang C, Xie Y, Song F, Zhou X: A tomato glutaredoxin geneSlGRX1regulates plant responses to oxidative, drought and salt stresses. Planta. 2010, 232 (6): 1499-1509. 10.1007/s00425-010-1271-1.
- 46.
Sagi M, Davydov O, Orazova S, Yesbergenova Z, Ophir R, Stratmann JW, Fluhr R: Plant respiratory burst oxidase homologs impinge on wound responsiveness and development inLycopersicon esculentum.Plant Cell. 2004, 16 (3): 616-628. 10.1105/tpc.019398.
- 47.
Guan LM, Scandalios JG: Hydrogen peroxide-mediated catalase gene expression in response to wounding. Free Radical Bio Med. 2000, 28 (8): 1182-1190. 10.1016/S0891-5849(00)00212-4.
- 48.
Guan LM, Zhao J, Scandalios JG:Cis-elements andtrans-factors that regulate expression of the maize Cat1 antioxidant gene in response to ABA and osmotic stress: H2O2可能中间信号分子吗the response. Plant J. 2000, 22 (2): 87-95. 10.1046/j.1365-313x.2000.00723.x.
- 49.
Mengiste T: Plant immunity to necrotrophs. Annu Rev Phytopathol. 2012, 50: 267-294. 10.1146/annurev-phyto-081211-172955.
- 50.
Jones JD, Dangl JL: The plant immune system. Nature. 2006, 444 (7117): 323-329. 10.1038/nature05286.
- 51.
Glazebrook J: Contrasting mechanisms of defense against biotrophic and necrotrophic pathogens. Annu Rev Phytopathol. 2005, 43: 205-227. 10.1146/annurev.phyto.43.040204.135923.
- 52.
AbuQamar S, Chai MF, Luo H, Song F, Mengiste T: Tomato protein kinase 1b mediates signaling of plant responses to necrotrophic fungi and insect herbivory. Plant Cell. 2008, 20 (7): 1964-1983. 10.1105/tpc.108.059477.
- 53.
Li X, Zhang Y, Huang L, Ouyang Z, Hong Y, Zhang H, Li D, Song F: Tomato SlMKK2 and SlMKK4 contribute to disease resistance againstBotrytis cinerea.BMC Plant Biol. 2014, 14: 166-10.1186/1471-2229-14-166.
- 54.
Benito EP, ten Have A, van’t Klooster JW, van Kan JAL: Fungal and plant gene expression during synchronized infection of tomato leaves byBotrytis cinerea.植物病理学研究。1998欧元,104(2):207 - 220。10.1023/A:1008698116106.
- 55.
Thordal-Christensen H, Zhang Z, Wei Y, Collinge DB: Subcellular localization of H2O2in plants. H2O2accumulation in papillae and hypersensitive response during the barley-powdery mildew interaction. Plant J. 1997, 11 (6): 1187-1194. 10.1046/j.1365-313X.1997.11061187.x.
Acknowledgements
This work was supported by the National High-Tech R & D Program (No. 2012AA101504 and No. 2012AA101505), the National Basic Research Program of China (2009CB119005) and the Research Fund for the Doctoral Program of Higher Education of China (20120101110070).
Author information
Affiliations
Corresponding author
Additional information
Competing interests
The authors declare that they have no competing interests.
Electronic supplementary material
Alignment of tomato SlSR protein sequences and positions of the VIGS fragments in the SlSR proteins.
Additional file 1: The GenBank accession numbers for SlSR proteins are as follows: SlSR1, ADK47999; SlSR1L, AEX31181; SlSR2, AEX07774; SlSR2L, AEX07775; SlSR3, AEX07776; SlSR3L, AEX07778; SlSR4, AEX07777. These SlSR protein sequences were deposited by Yang et al. [29]. Regions selected for the VIGS fragments of each of SlSR genes were underlined with red lines. (DOC 1 MB)
Sequences of the VIGS fragment for
Additional file 3:SlSRgenes.(DOC 34 KB)
Authors’ original submitted files for images
Below are the links to the authors’ original submitted files for images.
Rights and permissions
Open AccessThis article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made.
图片或其他第三方的材料在这rticle are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder.
To view a copy of this licence, visithttps://creativecommons.org/licenses/by/4.0/.
The Creative Commons Public Domain Dedication waiver (https://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
About this article
Cite this article
Li, X., Huang, L., Zhang, Y.et al.Tomato SR/CAMTA transcription factors SlSR1 and SlSR3L negatively regulate disease resistance response and SlSR1L positively modulates drought stress tolerance.BMC Plant Biol14,286 (2014). https://doi.org/10.1186/s12870-014-0286-3
Received:
Accepted:
Published:
Keywords
- Tomato (Solanum lycopersicum)
- SR/CAMTA
- Disease resistance response
- Drought stress